|
For more information on how these figures were generated, see CoolProp.Plots. If we don’t arrive back at the original T,P values, there is a problem in the flash routine in CoolProp. The balanced chemical equation for the combustion of dodecane is: C12H26 + 18. When dodecane is burned, it reacts with oxygen to produce carbon dioxide (CO2) and water (H2O). The heat of combustion of dodecane is approximately 44.7 MJ/kg. In this figure, we start off with a state point given by T,P and then we calculate each of the other possible output pairs in turn, and then try to re-calculate T,P from the new input pair. The chemical formula of dodecane is C12H26. The red curve is the maximum temperature curve, and the blue curve is the melting line if one is available for the fluid. A red + is a failure of the flash routine, a black dot is a success. 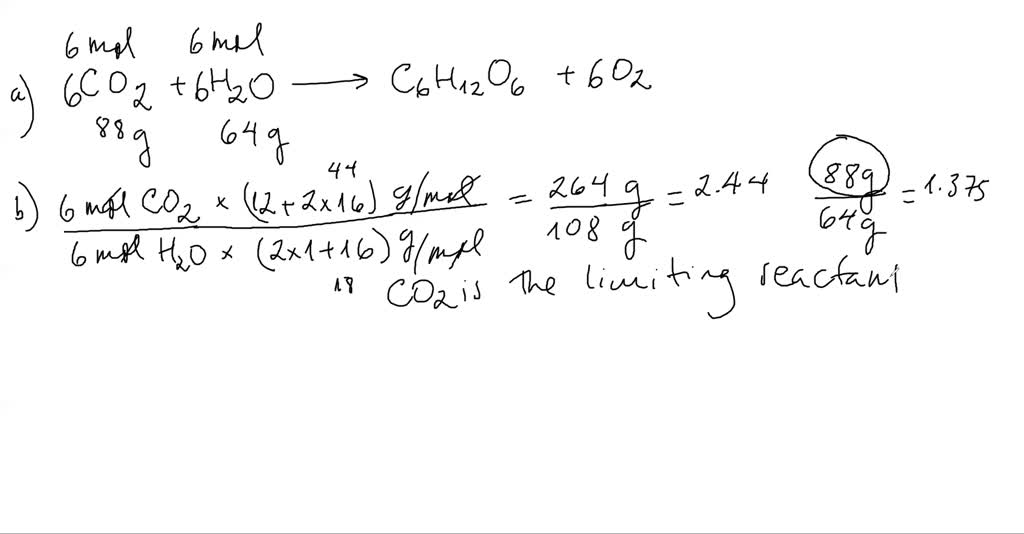
The following figure shows all the flash routines that are available for this fluid. You can also download this figure as a PDF. You can download the script that generated the following figure here: (link to script), right-click the link and then save as… or the equivalent in your browser. 
They are all results obtained in the form \(Y(T,\rho)\), where \(Y\) is the parameter of interest and which for all EOS is a direct evaluation of the EOS This figure compares the results generated from CoolProp and those generated from REFPROP. 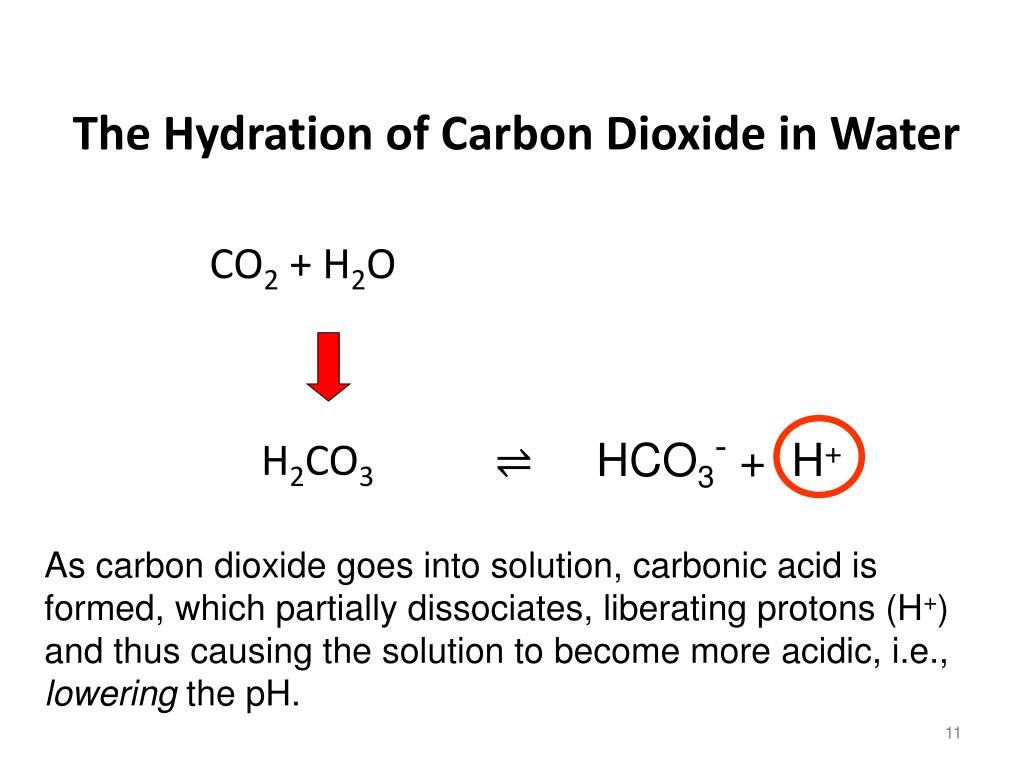
NDodecane, Dodecane, DODECANE, N-DODECANE, C12, NC12H26, n-C12H26 Fluid Information ¶ Use Hess’s Law (BLBMWS section 5.6) to calculate the heat of combustion of dodecane using heat of formation data that can be obtained from the National Institute of Standards and Technology (NIST) web site. Recommended Correlations for the Surface Tension of Common Fluids. Write the balanced equation for the combustion of dodecane (C12H26) with molecular oxygen gas to form carbon dioxide and water. Therefore the density variation decreases from 0.29 to 0. Surface Tension ¶Ī. Mulero, I. Cachadiña, and M. I. The maximum density variations in n-butane & carbon dioxide and n-dodecane & carbon dioxide mixtures are 2.01 and 2.28 kg/m 3 when the thermodiffusion is absent and 1.22 and 2.11 kg/m 3 when the thermodiffusion is present. Huber, Arno Laesecke, and Richard Perkins. Densities and viscosities were measured along 11 isotherms at temperatures from (298.15 to 548.15) K and pressures up to 10 MPa for n-dodecane, and over a temperature range of (303.15 to 548. Huber, Arno Laesecke, and Richard Perkins. A vibrating-wire apparatus was used to measure liquid density and viscosity simultaneously under high-temperature conditions. Sometimes there may be multiple compounds with that element on one side, so you'll need to use your best judgement and be prepared to go back and try the other options.N-Dodecane ¶ References ¶ Equation of State ¶Įric W. Step 2: Multiply coefficients for compounds to balance out each elementįor each element that is not equal, try to balance it by adding more of it to the side with less. Step 1: Count the number of each element on the left and right hand sides Dodecane is a straight-chain carbon with 12 carbon atoms. When using the inspection method (also known as the trial-and-error method), this principle is used to balance one element at a time until both sides are equal and the chemical equation is balanced. Calculate the volume of carbon dioxide gas that is produced. To be balanced, every element in C12H26 + O2 = CO2 + H2O must have the same number of atoms on each side of the equation. Write a balanced chemical equation, including physical state symbols, for the combustion of liquid decane into gaseous carbon dioxide and gaseous water suppose 0.490 kg of decane are burned in air at a pressure of exactly 1 atm and a temperature of 110 C. Write a balanced chemical equation, including physical state symbols, for the combustion of liquid dodecane into gaseous carbon dioxide and gaseous water. 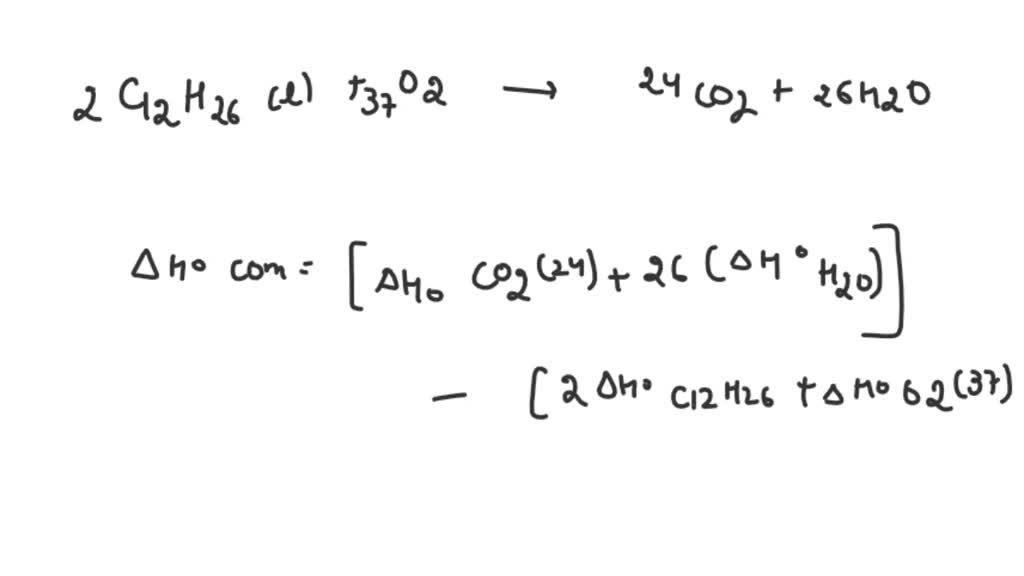
The law of conservation of mass states that matter cannot be created or destroyed, which means there must be the same number atoms at the end of a chemical reaction as at the beginning.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
